Identify the Effects of Ultra Weak Light on Alphacoronavirus and Vero Cells
Abstract
Coronavirus is causing many diseases and economic pain to humans and animals. The present study demonstrated how ultra weak lightcause changes in porcine epidemic diarrhea virus (PEDV) and Vero cells.
Ultra weak lightactivates Vero cells by lowering pH of maintain media, but have proven no effect of killing the virus.
Author Contributions
Academic Editor: Raul Isea, Fundación Instituto de Estudios Avanzados - IDEA, Hoyo de la Puerta, Baruta.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2021 Hee-Chun Chung, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Coronavirus is not only a problem for people like COVID-19, it causes a range of diseases in farm animals, some of which can be serious and are a threat to the farming industry 3. Economically significant coronaviruses of farm animals include porcine epidemic diarrhea virus (PEDV) and bovine coronavirus (BCoV), which both result in diarrhea in young age groups animals 4. PEDV has been circulating in Asia for several decades. It was identified for the first time during 1980s in Japan and China 8. PEDV became a global issue, especially in 2013, with a high death rate in the USA 9 Many researchers are working hard to get rid of PEDV, which has been a problem up until now 10.
In this study, the change was observed as a result of exposure of ultra weak light to cells and viruses respectively.
Methods
Ultra Weak Light
An optical apparatus (Photonia® manufactured by Biolight corporation) that emits light of ultra weak intensity was used, and its irradiance and optical spectrum were adjusted by passive optical components.
The measured irradiance at a position of 2 cm away from the center of the light emitting surface of the apparatus was about 5.2×10-9 W/cm2 and the dominant wavelength of the spectrum was 588 nm.
Virus (PEDV)
The PEDV strain DR-13 is a PEDV strain attenuated through serial passages resulting in a 51-nucleotide deletion in the ORF3 gene 7, and it is being used for a PED oral vaccine (DR13 106.5 TCID50/mL) commercialised in 2003 by Green Cross Vet. Prod. (South Korea).
PEDV Titration
Virus titration was carried out using a 48-well microplate with Vero cells. Virus cultures were serially diluted 10-fold with the virus replication medium containing trypsin. Confluent Vero cells of the microplate were washed three times with PBS and inoculated at 0.1 mL per well into five wells. Following adsorption for 1 h at 37 oC, the inocula were removed, and the cells were washed three times with PBS. Subsequently, 0.1 mL of fresh virus replication medium containing trypsin was transferred into each well, and the cells were further incubated for 5 days at 37 8C. Fifty per cent tissue culture infective doses (TCID50) were expressed as the reciprocal of the highest virus dilution showing cytopathic effect (CPE) 6 Antiviral effect of ultra weak light was expressed by inhibition titer, which was
calculated as follows: (log10(TCID50/ml of control group) – log10(TCID50/ml of experimental group)) 1
Ultra Weak Light Exposure to Light with Changes in Viral Titer
Set up an experimental group that exposed 10 ml of a pre-calculated (106.5 TCID50/ml titer) PEDV to ultra weak light and that has not been exposed to light (control group) within the CO2 5% incubator. Light exposure times are 10, 24, 48, 72 and 96 hours, respectively.
Test the Influence of Vero Cells on Ultra Weak Light
After incubation of approximately 1,000 Vero cells in 25T flask in the CO2 5% incubator, the change in the number and pH of the cells is measured in the light treated experimental group and the non-lighted control group. Light exposure times are 10, 24, 48, 72 and 96 hours, respectively.
Identification of the Inhibition of the PEDV in Vero Cells in Response to Ultra Weak Light
Incubate the 48 well plate Vero cells a day earlier and prepare an experimental group which exposed times (10, 24, 48, 72 and 96 hours) of ultra weak light and an unexposed control group.
The ultra weak light exposed experimental group after inoculation with the appropriate pre-calculate virus (titer; 105.3 TCID50/ml) was compared with the non-exposed control group.
Validation of Significance Through Statistical Analysis
All experiments were repeated 3 times. Statistical comparisons were performed using ANOVA tests (p < 0.05) within the SPSS program (version 15.0.0) (SPSS Inc., USA). An asterisk (*) denotes statistical significance among the mean data.
Results and discussion
Testing the Influence of PEDV on the Ultra Weak Light
We demonstrated changes in PEDV titers during exposure times for ultra weak light.
Following exposure to ultra weak light, the PEDV inhibition above the logarithmic reduction = log 0.1 (20.567%) from 10h to 72h was confirmed, but the statistical analysis was not significant (Figure 1). The effectiveness of disinfectant as a disinfectant announced a maximum reduced titer, in which the virus titer was reduced by at least 4 log10 2, 5 was not met to be an effective dilution factor.
Figure 1.Changes in PEDV titers according to ultra weak light exposure times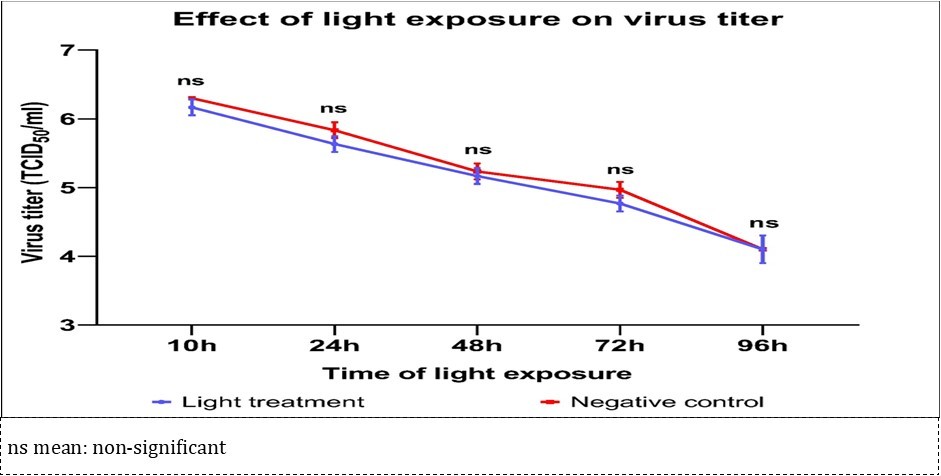
Testing the Influence of Vero Cells in Response to the Ultra Weak Light
We examined how Vero cells changes following exposure to ultra weak light.
After exposure to ultra weak light, the pH level maintained lower than the control group for 10 to 96 h, which enabled Vero cells, increasing the number of cells by 1.2 to 1.5 times more than the control group. In particular, differences due to larger effects were identified during light exposure for over 72 hours, indicating that the statistical analysis was also significant (Figure 2). The ultra weak light activated the cell by decreasing the pH of the growth medium.
Figure 2.Variation of Vero cell and pH due to exposure to ultra weak light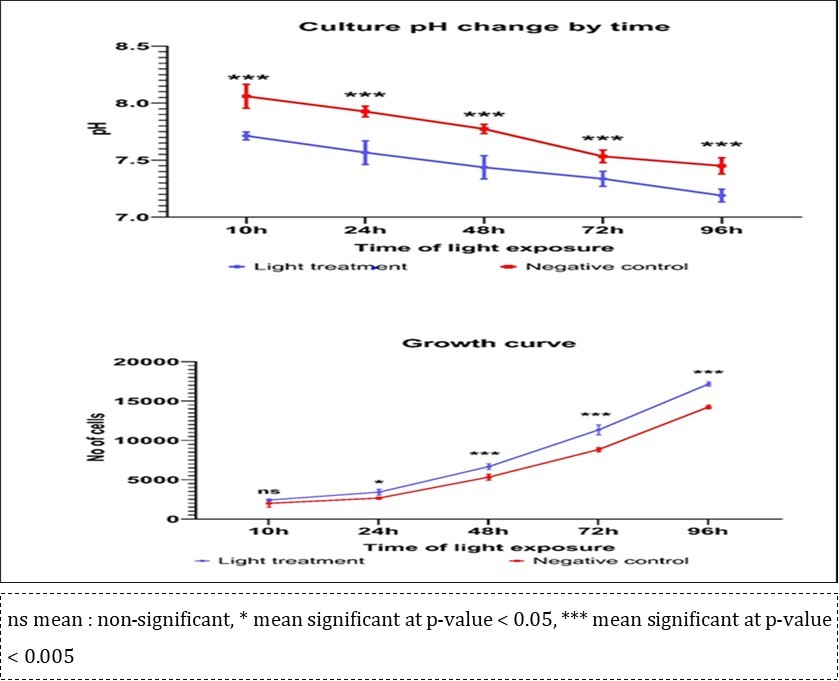
Identification of PEDV Inhibition in Vero Cells in Reaction to Ultra Weak Light
We found changes in PEDV in Vero cells exposed to ultra weak light.
After exposure to ultra weak light, the virus inhibition of log reduction=0.1 (20.567%) from 10h to 48h was confirmed, but statistical analysis showed no significant effects of virus reduction (Figure 3).
Figure 3.PEDV Inhibition in exposed ultra weak light within Vero Cells
Funding
This work was supported by the Korea Science and Engineering Foundation (KOSEF) grant funded by the Korea government (No. 2020R1I1A1A01054539) and by Korea Institute of Planning and Evaluation for Technology in Food, Agriculture and Forestry (IPET) through Technology Commercialization Support Program funded by Ministry of Agriculture, Food and Rural Affairs(MAFRA) (No. 821065-03), respectively.
Acknowledgments
The authors would like to thank Eun Ok Kim and Jung Ah Kim for their excellent technical assistance.
References
- 1.Chen Y-N, Hsueh Y-H, Hsieh C-T, Tzou D-Y, Chang P-L. (2016) Antiviral activity of graphene–silver nanocomposites against non-enveloped and enveloped viruses. International journal of environmental research and public health 13. 430.
- 2.Oliveira T M L de, I S Rehfeld, Guedes M I M C, Ferreira J M S, E G Kroon et al. (2011) Susceptibility of Vaccinia virus to chemical disinfectants. The American journal of tropical medicine and hygiene 85. 152.
- 3.Esudu E L. (2021) A survey on the impact of. Covid19 on Animal production and marketing. BusitemaUniversity .
- 4.Haake C, Cook S, Pusterla N, Murphy B. (2020) Coronavirus infections in companion animals: virology, epidemiology, clinical and pathologic features. , Viruses 12, 1023.
- 5.Jimenez L, Chiang M. (2006) Virucidal activity of a quaternary ammonium compound disinfectant against feline calicivirus: a surrogate for norovirus. , American journal of infection control 34, 269-273.
- 6.KUSANAGI K-i, KUWAHARA H, KATOH T, NUNOYA T, ISHIKAWA Y et al. (1992) Isolation and serial propagation of porcine epidemic diarrhea virus in cell cultures and partial characterization of the isolate. , Journal of Veterinary Medical Science 54, 313-318.
- 7.Park S-J, Moon H-J, Luo Y, Kim H-K, Kim E-M et al. (2008) Cloning and further sequence analysis of the ORF3 gene of wild-and attenuated-type porcine epidemic diarrhea viruses. , Virus genes 36, 95-104.
- 8.Song D, Park B. (2012) Porcine epidemic diarrhoea virus: a comprehensive review of molecular epidemiology, diagnosis, and vaccines. , Virus genes 44, 167-175.