Management of Plastıc Bronchıtıs in Chıldren: A Case Report and Lıterature Revıew
Abstract
Plastic bronchitis (PB) is a rare disease characterized by the presence of mucofibrinous plugs which may occlude and conform the shape of tracheobronchial tree. These casts are exteremely cohesive. Most common presenting symptoms include cough, fever and dyspnea and if the cohesive casts occlude the airway totally life-threatening complications and even death may occur. The aim of treatment is to remove the casts and adress the symptoms. In this study a 6-year-old boy with PB is presented. In addition to medical treatment obstructing casts were removed via rigid bronchoscope. It is aimed to review the clinical and radiographic features and choices of treatment in this disease.
Author Contributions
Academic Editor: Padmavathi Kora, Gokaraju Rangaraju Institute of Engineering & Technology, Hyderabad, India.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Volkan Sarper Erikci
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Plastic bronchitis (PB) or cast bronchitis is an uncommon and often underdiagnosed entity characterized by the presence of mucofibrinous plugs which may partially or completely occlude the airways 1, 2. These casts are exteremely cohesive and conform to the shape of tracheobronchial tree partially or totally. Cough, fever and dyspnea are the usual presenting symptoms in PB. However, if the cohesive casts occlude the significant portion of tracheobronchial tree life-threatening complications an deven death may occur. The etiology of PB has not been fully clarified and the aim of treatment is to remove the casts and adress the symptoms 3
In this study we present a 6-year-old boy with PB. In addition to medical treatment the problem of obstructing casts was solved with the aid of bronchoscopic removal of cohesive casts. It is aimed to review the clinical and radiographic features and choices of treatment in this rather rare entity under the light of relevant literature.
Case Report
A 6 year-old boy was admitted to our hospital with the symptoms of upper respiratory tract infection (URI). He had hemoptysis 2 days prior to admission to hospital. On admission he had respiratory distress, fever (39°). Plain chest graphy demonstrated a complete opacification of the left lung (Figure 1). Thorax ultrasonography was normal and high resolution computed tomography (HRCT) of the chest revealed a 50-60% narrowing of the left main stem bronchus starting from carina downwards (Figure 2). Although there was no history of foreign object aspiration an emergent bronchoscopy was performed for a possible unnoticed aspirated foreign body. A bronchial cast located at the carineal bifurcation of trachea measuring 3x1 cm was detected and easily removed with forceps during bronchoscopy (Figure 3). The material was whitish, rubbery in consistency. Histopathologic examination of the cast revealed that it consisted of condensed fibrin, neutrophils and eosinophils. Culture was also obtained from the specimen for a possible bacterial growth. Following bronchoscopy the patient required mechanical ventilation for ventilatory support in the mode of high frequency oscillatory ventilation (HFOV) with supplementery oxygen in intensive care unit (ICU). In his 3rd hospital day, control chest graphy of the child showed complete opacification of the left lung and for a chance of residual cast in the tracheobroncial tree a repeat bronchoscopy was performed. Two seperate casts from the left main stem bronchus measuring 1.5x1 cm and 2x1 cm in length were removed with forceps and lavage with aspiration of both right and left main stem bronchus was performed. Following second bronchoscopy the transcutaneous oxygen saturation increased from 77 to 99 and control chest roentgenogram showed equal expension of both lungs. While in his stay in ICU, in addition to mediacal treatment, respiratory physiotherapy was performed regularly. He was medically treated with aerosolized dexamethasone with adrenalin. Bacterial test obtained from cast was found positive for Streptococcus viridans and vancomycin was started. On his 5th day in ICU, his respiratory status worsened and a chest graphy showed a decrease in expension of the left lung and a control bronchoscopy was performed. In this third bronchoscopy trachea and both right and left main stem bronchus were found to be patent but were found to have excessive viscous secretions. Pulmonary toilet including lavage and aspiration of thick tenacious secretions was performed. On day 12 he was transferred from the ICU and 1 week later he was weaned off the ventilator. He underwent pulmonary rehabilitation. During his 3rd year of follow up he has no further episodes of respiratory distress and he is disease free.
Figure 1.Chest radiograph showing left-sided opacification.
Figure 2.HRCT of the patient showing narrowing of the left main stem bronchus starting from carina downwards.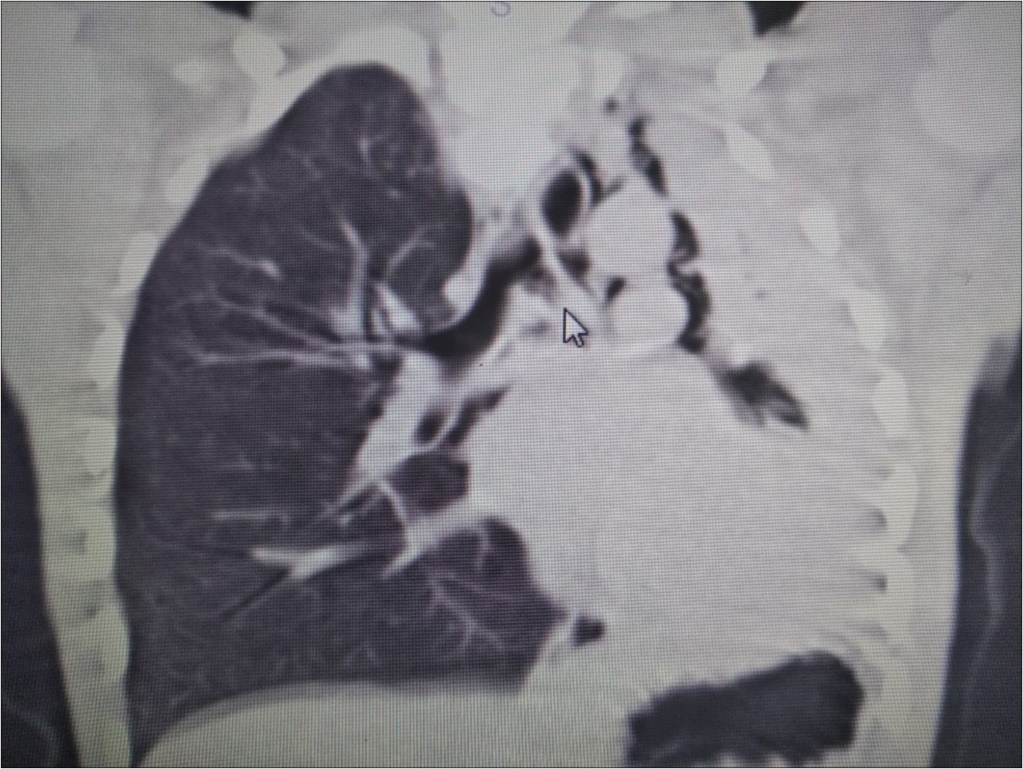
Discussion
Plastic bronchitis is a rare and severe respiratory disorder which is characterized by the formation of extensive gelatinous bronchial casts. Sometimes these casts conform the shape of the tracheobronchial tree 2. There is wide spectrum of presentation types including mild symptoms in one hand or life threatening disease with a mortality rate up to 60% especially in children with cardiopathies on the other 4, 5, 6, 7.
Description of this entity was documented as early as Galen in 131-200 A.D. 8, 9. First reports of PB dates back to the early 20th century and were associated with suppurative lung infections 10, 11. It was reported that 72 patients with PB were reported before 1989 12.
There are various classifications for casts reported in the literature. Seear et al. classified bronchial casts into two histopathologic groups 13. Type-1 casts were considered inflammatory comprising of dense fibrin structure and eosinophilia. Type-2 cases showed acellular histopathologic finding containing mucin and these cases had commonly congenital heart disease. Later Brogan et al. reclassified casts depending on the disease states of asthma, cardiac and idiopathic 14. In another further classification suggestion, the cases were categorized and expanded to cardiac, lymphatic, asthmatic and sickle-cell-related disease states 15.
The exact pathophysiology of PB is unknown. Increased pulmonary venous pressure, as seen in patients undergoing surgical intervention for Fontan procedure, may lead to abnormal response of respiratory epithelium producing excessive tenacious mucus may play a role 16, 17. Lymphatic dysfunction including elevated intrathoracic lymphatic pressure, possible lymphobronchial fistula and eventual endobronchial lymph leakage is another proposed mechanism of this disease 18. In another proposal, it has been suggested that inflammatory process as seen in activated asthma patients would cause dysregulation of formation of mucus resulting in tenacious casts which may obstruct tracheobronchial tree 15. Whatever the inciting mechanism, the end result is obstruction of respiratory tract partially or totally.
The prevalance of PB is unknown but a female predominance of cases has been reported 3. Clinical spectrum of disease varies greatly. Chronic cough, dyspnea and hemorrhage and/or hemoptysis, as in our case, are usually observed. Wheezing is the most common finding during physical examination. If the occluding cast obstructs the airway totally catastrophic situations such as suffocation and even sudden death may be observed.
Radiographic findings are nonspecific and include atelectasis secondary to airway obstruction, hyperinflation due to air trapping, unilateral opacification, as seen in our case, bronchiectasis and lung infiltrates. It has been reported that HRCT may allow in some cases the visualization of bronchial casts in the airways of larger caliber 19. However the gold standard in diagnosing PB is bronchoscopy which reveals the airway obstruction by BC 3.
PB is usually a complication of an underlying disease and definitive therapy in these cases necessitates the treatment of this underlying condition in order to eliminate the formation of BC. Underlying diseases are depicted in Table 1. It has been reported that PB is seen in children after 1-4% of Fontan surgery 20.
Table 1. Underlying diseases in patients with PB*.| Pneumonia/Eosinophilic pneumonia |
| Bronchiectasis |
| Asthma |
| Allergy |
| URI |
| Sickle cell acute chest syndrome |
| Acute myelogenous leukemia |
| Pulmonary lymphatic abnormalities |
| Pulmonary infection with Influenza A virüs |
| Toxic inhalation (chemicals or thermals) |
| Systemic lymphatic abnromalities (Turner’s and Noonan’s syndrome) |
| Bronchopulmonary aspergillosis |
| Cystic fibrosis |
| Chronic obstructive pulmonary disease |
| Pericarditis |
| DORV with PVA |
| RAD |
| Tricuspid atresia |
| Tetralogy of Fallot |
The aims of treatment in PB are to assist removal of BC from the respiratory tract of affected patients and measures to prevent and eliminate the formation of BCs. Initial management include bronchodilators, steroids, inhaled hypertonic saline, mucolytics and antibiotics. Topical fibrinolytic treatment aims to lysis of mucus plugs and removal with forced expectoration or removal with bronchoscopy. Of the topical agents reported in the treatment of these cases, inhaled heparin, urokinase, tissue plasminogen activator (tPA) act on fibrin, recombinant DNAse acts on cellular material and nebulized N-acetyl cystein acts on mucin 20. According to Brogan et al. acetylcystein has the greatest effect in dissolving these casts over DNAse, urokinase and tPA 14.
The use of bronchoscopy in removal of BC in patients with PB is first reported by Raghuram et al. in 1997 21. It may be performed using flexible or rigid material. Rigid bronchoscopy has the advantage of being both diagnostic and therapeutic and was used in the presented case for removal of obstructing BC. It has been reported that if friability of plugs makes the bronchoscopic forceps removal impossible, rigid suctioning together with bronchoalveolar lavage during bronchoscopy may provide adequate clearance of BCs from the airways of affected patients 22.
In conclusion, PB is a challenge for the first liners of medical providers either in diagnosis or in treatment. Being an extremely rare and sometimes fatal disease, clinicians should have a high index of suspicion of PB in cases with right clinical setting. Prompt intervention including bronchoscopic removal of casts together with medical treatment is paramount. As our understanding of regulation of mucus production improves new treatment modalities may allow definitive therapy for patients with PB.
References
- 1.Walker P A, Shah S K, Letourneau P A. (2013) Treatment of plastic bronchitis using serial flexible bronchoscopy and aerosolized heparin therapy. , Eur J Pediatr Surg 23, 157-60.
- 2.Hasan R A, Black C, Reddy R. (2012) Plastic bronchitis in children. Fetal and Pediatric Pathology. 31, 87-93.
- 3.Eberlein M H, Drummond M B, Haponik E F. (2008) Plastic bronchitis: a management challenge. , Am J Med Sci 335, 163-9.
- 4.Nayar S, Parmar R, Kulkarni S, Cherian K M. (2007) Treatment of plastic bronchitis. Ann Thrac Surg. 83, 1884-6.
- 5.Sanerkin N G, Leopold J G. (1966) Plastic bronchitis, mucoid impaction of the bronchi and allergic bronchopulmonary aspergillosis, and their relationship to bronchial asthma. Ann Allergy. 24, 586-94.
- 6.Bowen A D, Oudjhane K, Odagiri K, Liston S L, Cumming W A et al. (1985) Plastic bronchitis: large, branching, mucoid bronchial casts in children. , Am J Radiol 144, 371-5.
- 7.Jett J R, Tazelaar H D, Keim L W, Ingrassia T S. (1991) Plastic bronchitis: an old disease revisited. Mayo Clin Proc. 66, 305-11.
- 10.Bettman M. (1902) Report of a case of fibrinous bronchitis with a review of all cases in the literature. , Am J Med Sci 123-304.
- 11.Mulligan P B, Spencer R D. (1924) Chronic fibrinous bronchitis as a symptom of mediastinal compression. , JAMA 82, 791-2.
- 12.Hoskison E, Judd O, Dickinson E, Dux A. (2010) Plastic bronchitis requiring bronchoscopy. , J Laryngol Otol 124(12), 1321-4.
- 13.Seerer M, Hui H, Magee F. (1997) Bronchial casts in children: a proposed classification based on nine cases and review of the literature. , Am J Respir Crit Care Med 155, 364-70.
- 14.Brogan T V, Finn L S, Pyskaty D J, Redding G J, Ricker D et al. (2002) Plastic bronchitis in children: a cses series and review of the medical literature. Pediatric Pulmonology. 34, 482-7.
- 15.Madsen P, Shah S A, Rubin B K. (2005) Plastic bronchitis: a new insights and classification scheme. Paediatr Respir Rev. 6(4), 292-300.
- 16.Silva R C, Simons J P, Chi D H. (2011) Endoscopic treatment of plastic bronchitis. Arch Otolaryngol Head Neck Surg. 137, 401-3.
- 17.Do P, Randhawa I, Chin T, Parsapour K, Nussbaum E. (2012) Successful management of plastic bropnchitis in a child post Fontan: case report and literature review.Lung. 190(4), 463-8.
- 18.Languepin J, Scheinmann P, Mahut B. (1999) Bronchial casts in children with cardiopathies: the role of pulmonary lymphatic abnormalities. Pediatr Pulmonol. 28, 329-36.
- 19.Peixoto A O, Riberio J D, da Costa RM, FAL Marson. (2017) Plastic bronchitis: a case report. Glob L Allergy. 3(2), 22-6.
- 20.Singhi A K, Vinoth B, Kuruvilla S, Sivakumar K. (2015) Plastic bronchitis. Ann Pediatr Card. 8, 246-8.

