Beneficial Impacts of Solanum aethiopicum L. in Diabetes Control
Abstract
Garden egg nutritional values are numerous. However, few nutritional and epidemiological data exist on the impacts of garden egg consumption on diabetes control. In this experimentally-control designed nutritional study, the effects of scarlet garden egg species (Solanum aethiopicum L.) on lipoglycemic profile, weight control and, pancreas histoarchitecture in diabetic male Wistar rats were examined. Twenty-One adult male Wistar rats inducted with diabetes were randomly categorized into three groups (n = 7, each): Diabetic control (DC); Diabetic treated with aqueous extract of garden egg (DEE) and Diabetic fed with garden egg-supplemented diet (DSE). Animals were fed for six weeks according to the experimental design. Glycemic status and body weights were assessed twice weekly while lipid analysis was conducted at the entry and 6th week of the study. Oral glucose tolerance (OGT) test was conducted. Gross analysis and tissue histology of the pancreas were assessed by Hematoxylin and Eosin (H&E) staining technique. Statistical analysis was done using analysis of variance, and the results were expressed as mean ± S.E.M. at P < 0.05. Garden egg reduced mean body weight gain (DSE: 14.53%; DEE: 10.58%; P value = 0.04) and decreased blood glucose concentrations (DEE: 37.33%; DSE: 18.68%; P = 0.03) with corresponding improved lipid profile, glycemic tolerance and control (DEE > DSE) and, preserved pancreas histoarchitecture in diabetic Wistar rats. Solanum aethiopicum (garden egg) consumption (as fresh fruit or supplemented diet) preserves pancreatic tissue histoarchitecture and improves lipoglycemic profile and weight control in diabetic Wistar rats.
Author Contributions
Copyright © 2022 Magnus Michael Chukwudike Anyakudo, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Diabetes mellitus is a chronic metabolic disorder of glucose metabolism with alarming incidence and mortality resulting from the associated risks and complications [1]. Diabetics have a constant challenge in maintaining appropriate blood sugar level while self-discipline and care are indispensable for increase life expectancy. Diabetes management required a holistic approach to care to prevent both acute and long-term complications. In this regard, nutritional management plays a significant role as pathophysiological basis of diabetic complications unravels. Many patients succumbed to diabetic complications due to its associated astronomical financial burden. To achieve adequate glycemic control, nutrition therapy is vital for both diabetic and prediabetic patients. Therefore, urgent interventional measures with less economic constraints are necessary to alleviate the associated morbidity and mortality.
The use of diets in the treatment of diabetes has long been established as a chief corner stone among other management modalities. Investigating into our locally available and affordable food crops of medicinal values and, their incorporation into diabetic menu is a central focus of our research development in diabetes control. In this study, the beneficial values of a species of garden egg (Solanum aethiopicum L.) in diabetes control were explored. The scarlet eggplant (Solanum aethiopicum L.; family: Solanaceae) is an indigenous vegetable species grown in African countries. S. aethiopicum is the second most widely cultivated eggplant, occurring in Africa, and in some parts of the Caribbean, Brazil and southern Italy 2, 3.
Garden egg (also known as eggplant) comes in varieties of species, shape, color and, taste valued for its bitterness. Figure 1 depicts some varieties of Solanum aethiopicum L. cultivars. Taxonomically, four cultivars of S. aethiopicum have been recognized including Gilo, Shum, Kumba and Aculeatum 4, 5.
Figure 1.Some morphotypes of Solanum aethiopicum L. cultivars
It is an edible, spongy and, absorbent fruit (berry) grown worldwide and used as a food crop in several countries in Africa 6, 7. In various parts of the world, eggplant has different nomenclature. In Nigeria, eggplant is called ‘Igbagba’ by the Yorubas, ‘Gauta’ by the Hausas and ‘Afufa’ or ‘Anara’ by the Ibos 8. Apart from its low calories and high moisture contents, phytochemical analysis of eggplant shows that it is a rich source of various essential organic and inorganic compounds such as histidine, nasunin, oxalic acid, anthocyanin, chlorogenic acid, solasodine, aspartic acid, tropane, flavonoids, lanosterol, gramisterol, steroid alkaloids, glycoalkaloids, ascorbic acid and tryptophan that are present in fruits and leaves 9, 10, 11, 12, 13. Solanum aethiopicum contains essential nutrients such as carbohydrates, proteins, fat, fibers and vitamins that are vital for human health 14, 15. It has been shown to have several medicinal values and sometimes said to be neutraceutics 16, 17.
Aside nutritional and agricultural importance, numerous therapeutic benefits have been ascribed to garden egg due to its constituent phytonutrient compounds. Research has revealed that eggplant extracts possessed anticarcinogenic 18, 19, 20, antioxidants 21, 22, antiobesity 10, 23, anti-inflammatory 24, antidiabetic, antibacterial, antiulcer 25, 26 and, beneficial ophthalmic 27 properties. Fiber and phytonutrients contents present in eggplant have also been reported to help in digestion, cell membrane protection and immune and memory boosting 20.
Glycemic control in diabetes management has become an important goal of diabetes care which involves normalization of blood glucose levels coupled with improved quality of life, minimization of medications and, increase life expectancy. Therefore, this study investigated the beneficial impacts of Solanum aethiopicum (scarlet garden egg) on diabetes control by determining its effects on weight gain, lipoglycemic profile and control, as well as its effect on pancreas tissue histoarchitecture with the rationale to provide alternative dietary measure that is affordable, accessible and available for euglycemic control of diabetes in affected individuals (especially people of low socioeconomic class) constrained by the financial burden of conventional diabetes management.
Materials and Methods
The scarlet garden eggs (Solanum aethiopicum) were purchased from a local market in Ondo State metropolis with the help of a botanist who assisted in the identification of the species. After removal of the calyx and proper washing and cleansing of the eggplant with distilled water, it was allowed to air-dry and weighed. 300g of the washed and air-dried garden egg was homogenized with 300 ml of distilled water to a fine and smooth texture mixture using a laboratory blender. The extract obtained through filtration using fine muslin cloth was collected in a container and kept at 4°C in the refrigerator. Portions used for oral administration to assess glycemic impacts were prepared fresh on weekly basis.
The compositions of the control and the garden egg-supplemented diets were based on the standard diet formulas used for assessing weight gain in rodents during commercial feeding studies (Table 1).
Table 1. Composition of control and test diets| Nutrient components | Ingredients used | Normal diet (% per 100g of feed) | Garden egg-supplemented diet (% per 100g of feed) |
| Carbohydrates | Maize | 40 | 40 |
| Wheat offal | 15 | 15 | |
| Fat and Oil | Palm kernel cake | 20 | 20 |
| Groundnut cake | 10 | 10 | |
| Soya bean meal | 10.5 | 10.5 | |
| Protein | Oyster shell | 1.0 | 1.0 |
| Bone meal | 3.0 | 3.0 | |
| Vitamins | Growth premix | 0.25 | 0.15 |
| Mineral Salt | Salt | 0.25 | 0.25 |
| ADDITIVES | Garden egg | - | 0.1 |
| Total | 100 | 100 |
Inthis study, twenty-one adult male Wistar rats (Rattus norvegicus) weighing 180-220g were used. The animals were acclimatized for two weeks in a well-ventilated animal house inside polypropylene cages with stainless wire mesh top at normal and standard laboratory conditions of temperature and relative humidity. During this period, animals were fed with standard rat chow (Ladokun feeds Nig. Ltd.) and water ad libitum. Under each cage, replaceable numbered blotters papers were placed to catch the spilled diet that was measured to make up for the daily serving ration. Weights of the rats were measured twice per week.
Using freshly prepared alloxan monohydrate (Kermel chemicals, China) solution administered intraperitoneally at a dose of 150 mg/kg body weight, the animals were inducted diabetic after 12hrs of overnight fast with free access to water. Four days later, diabetes was confirmed using glucometer (Fine Test Blood Glucose Monitoring System, OSANG Healthcare Co., Ltd., Korea). Rats with fasting blood sugar (FBS) concentrations above 150 mg/dL were considered diabetic and randomly selected into three groups according to the study design.
Following induction, animals were randomly categorised into three groups (n = 7/group) as follows:
Group diabetic control (DC)
Group diabetic treated with garden egg aqueous extract (DEE)
Group diabetic treated with garden egg supplemented diet (DSE)
To evaluate the effects of garden egg on glycemic status and tolerance, body weight and, lipid profile, groups DC and DEE rats were fed with control diet throughout the study period while DSE rats were fed with garden egg-supplemented diet only. In addition, DEE rats were treated with garden egg aqueous extract administered orally by flexible orogastric cannular. Food and water intake were monitored twice daily while glycemic status and body weight were assessed twice a week. Animal handling procedures and the experimental protocols were executed in strict compliance with the internationally accepted ethics, principles and, guidelines for laboratory animal use and care in research 28. The study was ethically reviewed and approved by the Institution Review Committee (UNIMEDTH/REC/28012022)
Blood samples used for blood glucose concentration and lipid profile assays were obtained from the tail veins (via phlebotomy) and the heart (via cardiac puncture) respectively. The blood samples for the lipid profile analysis were collected in sample bottles containing tripotassium (K3) Ethylene Diamine Tetraacetic Acid (EDTA) anticoagulant while the blood for glucose concentration assessment was applied directly to the glucometer strip.
After overnight fast, oral D-glucose load of 2gm per kg body weight (dissolved in distilled water) was administered via flexible orogastric cannula to all rats at the end of the 6th week. Blood samples were then taken at 30 minutes interval from the tail vein to determine glucose concentrations for a period of 2 hrs.
The lipid profile assay was conducted both at the beginning and the end of the study. Enzymatic colorimetric method was used to measure the lipid parameters (TC, TG, HDL) using a dry-chemical automatic analyzer AU-5200 OLYMPUS (Randox Laboratories, San Francisco, USA). LDL level was determined by the Friedewald formula 29 as follows:
VLDL (mg/dL) = TAG/5
LDL (mg/dL) = TC - VLDL – HDL
Following light anaesthesia with ethyl ether in a glass dome, animals were dissected to harvest the pancreas which was weighed and then histologically processed using animal tissues technique protocol and then examined under light microscope after H&E staining.
With appropriate statistical method and program of Microsoft Excel and SPSS, data obtained were analyzed. Results (all the mean values) are expressed as group mean ± SEM (standard Error of Mean). Comparison between groups was made using one way analysis of variance (ANOVA) followed by Duncan’s multiple range tests. Significance level was set to P < 0.05.
Results
Table 2 depicts the initial and final mean body weights for each group. Consumption of S. aethiopicum (scarlet garden egg) caused a significant decrease in mean body weight gain in both DSE (14.53%) and DEE (10.58%) rats compared with the control (DC) rats (18.34%). The difference in the mean weight gain between the DSE and DEE was significant (P = 0.04) indicating that, consumption of fresh or raw garden egg, impacts better weight-lowering effect than when incorporated in mixed meal via cooking, boiling, frying or any other process.
Table 2. Effect of S. aethiopicum on body weight and weight gain (n = 5/group)| Parameters | Experimental Diabetic Animal Groups | ||
|---|---|---|---|
| Control DC | Garden Egg-Supplemented diet DSE | Garden Egg Extract-Treated DEE | |
| Final Mean Weight (g) | 220.4 ± 6.47 | 214.4 ± 2.78 | 204.8 ± 2.59 |
| Initial Mean Weight (g) | 186.2 ± 9.15 | 187.2 ± 4.22 | 185.2 ± 5.63 |
| % Mean Weight Gain | 18.34% | 14.53*% | 10.58% |
The hypoglycemic effect of S. aethiopicum on venous mean FBS concentrations over the period of study in experimental diabetic rats is shown in Table 3. A significant (P = 0.032) reduction in mean FBS concentrations was observed in DEE rats and DSE rats compared with the control. The observed difference in the mean FBS values between DSE and DEE groups was comparably significant (P = 0.025). In a similar manner to its effect on body weight, S. aethiopicum consumption in fresh form potentiates more beneficial antidiabetic effect on glycemic status than when incorporated in mixed meal in diabetic rats. Overall, consumption of this garden egg species either as fresh or in supplemented form, impacts good glycemic control in diabetic rats.
Table 3. Effect of S. aethiopicum on venous mean FBS concentrations (n = 5/group)| Parameters | Experimental Diabetic Animal Groups | ||
|---|---|---|---|
| Control DC | Garden Egg-Supplemented Diet DSE | Garden Egg Extract DEE | |
| Final Mean FBS (mg/dL) | 211.6 ± 7.2 | 164 ± 3.6 | 188.6 ± 19.6 |
| Initial Mean FBS (mg/dL) | 178.6 ± 2.3 | 201.6 ± 25.9 | 259 ± 36.7 |
| % Change in Mean FBS | 18.48% | -18.68*% | -37.33**% |
Figure 2 illustrates the effect of S. aethiopicum on glycemic tolerance as assessed by the incremental areas under the glycemic response curves. Garden egg significantly enhanced glycemic tolerance in DSE and DEE rats compared with the controls. The glycemic tolerance effect on DEE rats is comparably better and more improved over that of the DSE rats. The glycemic response to glucose load in all experimental groups peaked at 30 minutes of the 2 hour-duration.
Figure 2.Effect of S. aethiopicum on glycemic tolerance/profile (n = 5/group).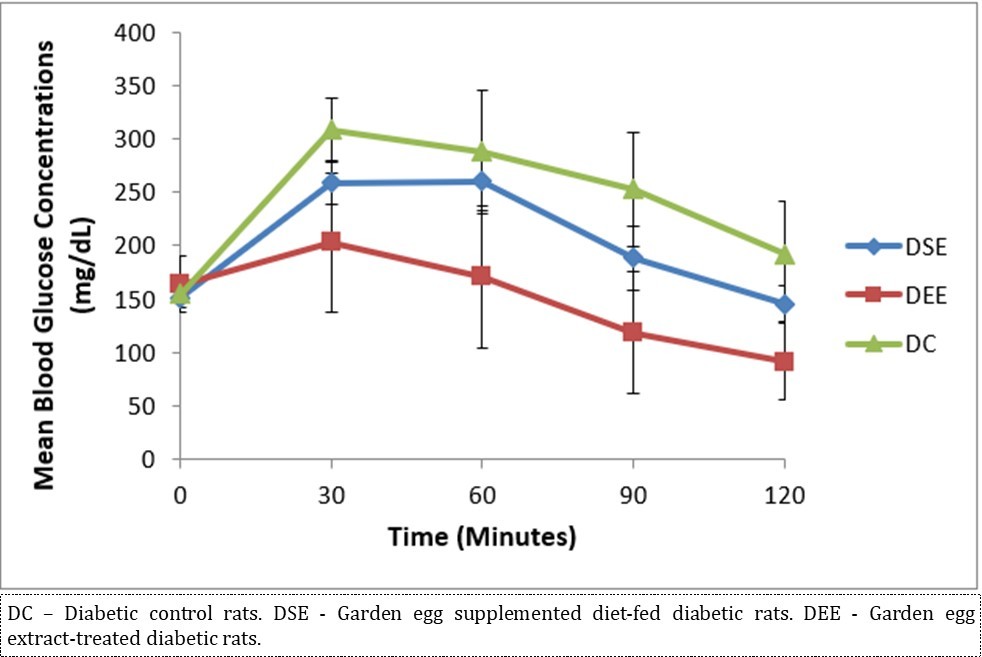
Effect of S. aethiopicum consumption on serum lipid parameters is expressed in Figure 3. At the end of the study, it caused significant (P < 0.05) decrease in TC, TG and LDL concentrations with corresponding significant increase in HDL concentration in DSE and DEE rats (DEE > DSE) compared with DC. This study reveals that consumption of S. aethiopicum improves lipid profile in diabetic rats.
Figure 3.Effect of S. aethiopicum on lipid profile in diabetic Wistar rats (n = 5/group)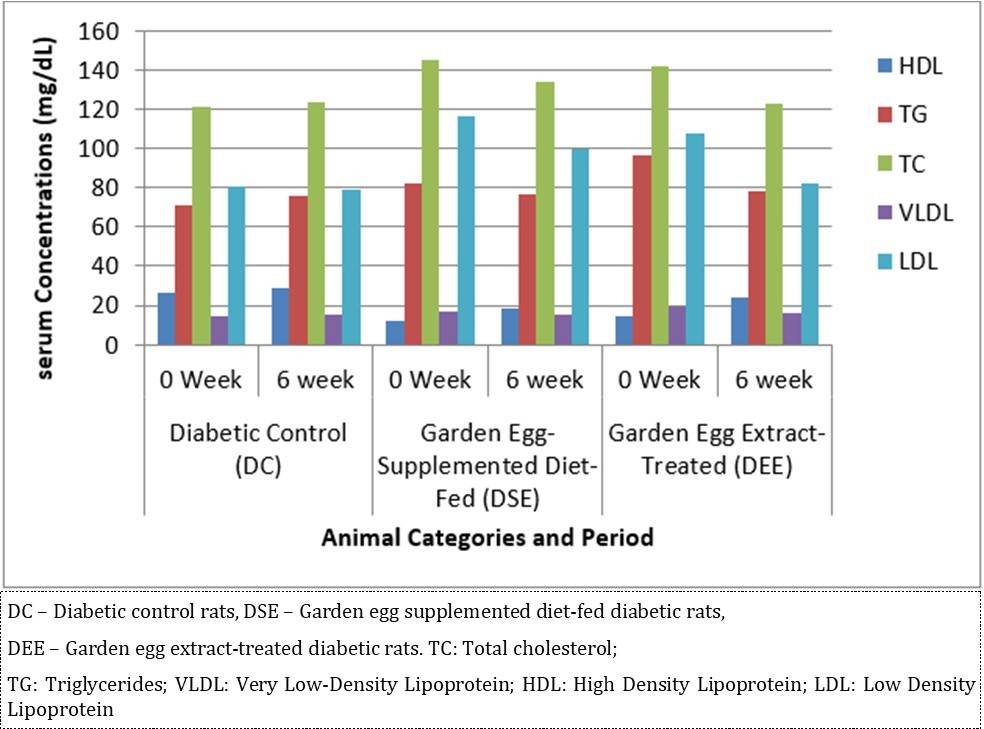
No comparable significant change observed in the mean weights of the extracted pancreas. The photomicrographs (Figure 4, Figure 5, Figure 6 and Figure 7) of the pancreatic tissue histology revealed preserved histoarchitecture in DSE and DEE groups as compared with the normal appearance of pancreas histoarchitecture from a normal healthy Wistar rat. Photomicrograph of the pancreas from diabetic control (DC) rats (Figure 7) shows distorted Islet margin with lymphocytic cells infiltration and moderate necrotic parenchyma.
Figure 4.Normal Photomicrograph of the pancreas from a non-diabetic healthy rat demonstrating normal histoarchitecture (H &E stain X400 magnification)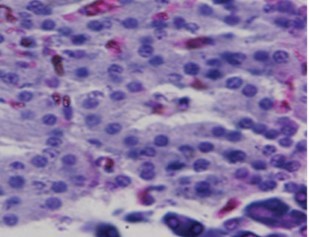
Figure 5.Photomicrograph of the pancreas from DEE rats showing preserved normal pancreas histoarchitecture (H &E stain X400 magnification)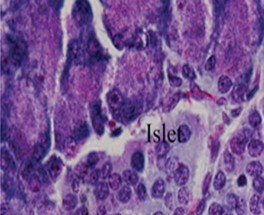
Figure 6.Photomicrograph of the pancreas from DSE rats showing preserved normal pancreas histoarchitecture (H &E stain X400 magnification)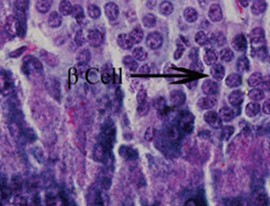
Figure 7.Photomicrograph of the pancreas from diabetic control (DC) rats showing distorted Islet margin (arrow) with lymphocytic cells infiltration and moderate necrotic parenchyma (H &E stain X400 magnification)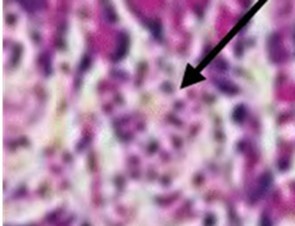
Discussion
The beneficial dietary impacts of S. aethiopicum consumption in diabetes control were evaluated in this experimentally-control designed nutritional study which lasted six weeks. In this study, the scarlet garden egg was administered in two forms (supplementation and extract) and its effects on body weight gain; glycemic status, tolerance and control; pancreas histoarchitecture and, serum lipid parameters were investigated while the influence of the mode of administration on the investigated parameters was also examined. Findings obtained revealed that S. aethiopicum possessed organoprotective, antiobesity, antidiabetic and antilipidemic potentials important in diabetes control. This study also revealed that consumption of S. aethiopicum in raw form as fresh fruit impacts better lipoglycemic control than when ingested in mixed meal as supplemented diets. Healthy weight loss is a goal for many patients specifically those confronting obesity while diet, physical activity, medications, and bariatric surgery all have varying roles in helping patients to achieve healthy weight loss. In this study, consumption of S. aethiopicum either as extract or supplemented significantly reduced weight gain in diabetic rats. However, the weight-lowering effect is more pronounced with the extract (Table 2). This observation implies that, eating scarlet garden egg as fresh fruit confers adequate and optimal weight-lowering benefit than when supplemented in diet as seen in various traditional cuisines such as boiling with yams, frying with eggs, and pounding with groundnut to make sauce, soup and so on. This reduction in weight by S. aethiopicum may be attributed to its high fibre and low cholesterol and protein contents 20 which prevent rapid weight gain and irrational eating.
Due to different species of garden egg in nature, likelihood of variation in its effect on weight reduction is expected. There is an uncited claim by some nutritionists that green garden egg species works better for weight loss compared to other species. However, findings obtained in our study using the white species with green stripes (S. aethiopicum L) revealed a remarkable reduction in weight gain in diabetic rats. Though, our present study did not compare species effect on weight gain, in our subsequent research however, we shall conduct a study to correlate species and weight-lowering effect. Meanwhile, based on the present outcome of this study, recommendation of the white-with-green-striped species should be encouraged for beneficial optimal effect on weight reduction. The beneficial impacts of S. aethiopicum on glycemic status and profile are depicted in Table 3 and Figure 2. The mean FBS concentrations decreased significantly (P < 0.05) in both DEE and DSE groups (DEE >> DSE, P = 0.025) compared with the control. The observed difference in their values was similar in manner to its effect on weight gain. The glycemic tolerance improved remarkably in both groups. This antidiabetic potential may be attributed to a major phenolic compound called chlorogenic acid (5-O-caffeoyl-quinic acid; CGA) present in garden egg 13, 21. Bhaskar 13 and Plazas 21 studies revealed that chlorogenic acid possessed anti-obesity, anti-inflammatory, anti-diabetic and, cardio-protective properties. The incremental areas under the glycemic response curves of DEE and DSE rats decreased remarkably compared with the control (Figure 2). The postprandial glycemic response peaked at 30 minutes of glucose challenge in both groups. The TC, TG and LDL-C concentrations decreased significantly with corresponding significant increase in HDL concentration in DEE and DSE rats (Figure 3). However, the extract impacts more beneficial antilipidemic effect than the supplemented diet. This finding also suggests that the optimal antilipidemic effect can be derived when S. aethiopicum is consumed in fresh or raw form than in mixed meal. The antilipaemic effect of eggplants has been attributed to an important phytonutrient compound called anthocyanin found in abundance in the skin of the eggplant which was reported to improve lipid profile through lipid peroxidation and serum antioxidants concentrations increase 27. Besides this antilipaemic activity, anthocyanin has also been reported to play significant role against diabetes, neuronal problems, cardiovascular disorders, and cancer as well 30. The pancreatic tissues sections’ photomicrographs examined under high power magnification (x 400) light microscope revealed the effects of S. aethiopicum on pancreas histoarchitecture. Figure 4 shows the normal appearance of the pancreatic histoarchitecture obtained from a healthy non-diabetic Wistar rat. Comparison of the experimental grouped rats photomicrographs with the normal revealed that the histoarchitecture of the pancreas in DEE and DSE (Figure 5, Figure 6) rats were preserved with no visible pathologic changes while the photomicrograph of the pancreas from diabetic control (DC) rats (Figure 7) shows distorted Islet margin with lymphocytic cells infiltration and moderate necrotic parenchyma. Examining the histological consequence of any product prior to recommendation is very essential because histological analysis of target organs for pathological changes surveillance is necessary to enhance screening assessment of quality and safety of product prior to recommendation for human consumption 31
The aim of diet therapy in diabetes management is to achieve normoglycemia, maintain ideal body weight and improve lipid profile. Thus, dietary advice is geared towards averting symptoms of hyper and hypoglycemia and to eliminate or postpone secondary complications which may arise. Unfortunately, many people are yet to discover the secret of nutritional values of garden eggs which are numerous. Thus,for optimal health benefits and dietary control of diabetes, consumption of S. aethiopicum in raw or fresh form should be encouraged and advocated in diabetic individuals as revealed by this study.
Conclusion
Solanium aethiopicum (scarlet garden egg) consumption preserves pancreatic tissue histoarchitecture and improves lipoglycemic profile and weight control in diabetic Wistar rats. Findings of this study potentiate the beneficial impacts of S. aethiopicum in diabetes control. Therefore, recommendation of S. aethiopicum (which is cheap, available and accessible to the common man) for consumption in diabetic condition (either as fresh fruit or diet-supplemented) should be advocated to avert complications and risks of uncontrolled diabetes possibly resulting from financial constraints of conventional diabetes management.
Funding
This research received no external funding.
Acknowledgements
No acknowledgement.
References
- 1.Saeedi P, Petersohn I, Salpea P. (2019) Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Research and Clinical Practice. 157, 1-10.
- 2.Sunseri F, Alba Polignano G B, C V Lotti, Bisignano V, Mennella G. (2010) Genetic diversity and characterization of African eggplant germplasm collection Afr. , J. Plant Sci 4, 231-241.
- 3.Gramazio P, Blanca J, Ziarsolo P, F J Herraiz, Plazas M et al. (2016) Transcriptome analysis and molecular marker discovery in Solanum incanum and S. aethiopicum, two close relatives of the common eggplant (Solanum melongena) with interest for breeding. , BMC Genom 17, 1-17.
- 4.R N Lester, M C Daunay. (2003) Diversity of African vegetable Solanum species and its implications for a better understanding of plant domestication Schriften zu. , Genetischen Ressourcen 22, 137-152.
- 5.Horna D, Timpo S, Gruère G. (2007) Marketing underutilized crops: the case of the african garden egg (Solanum aethiopicum) in Ghana Global Facilitation Unit for Underutilized Species (GFU). 1-45.
- 6.Krishnaiah D, Sarbatly R, Nithyanandam R. (2011) A review of the antioxidant potential of medicinal plant species. , Food of Bioproducts Processing 89, 217-233.
- 7.Tindall H D. (2017) Fruit and vegetable in West Africa – digitized. , Journal of Plant Production and Protection 5, 34-45.
- 8.Igwe S A, Akunyili D N, Ogbogu C. (2003) (2003)Effects of Solanum melongena (garden egg) on some visual functions of visually active Igbos of Nigeria.J. Ethnopharmacol. 86, 135-8.
- 9.Prohens J, Whitaker B D, Plazas M. (2013) Genetic diversity in morphological characters and phenolic acids content resulting from an interspecific cross between eggplant, Solanum melongena, and its wild ancestor (S. , incanum). Ann. Appl. Biol 162, 242-257.
- 10.Casati L, Pagani F, Braga P C. (2016) Nasunin, a new player in the field of osteoblast protection against oxidative stress. , J. Funct. Foods 23, 474-484.
- 11.Linnewiel-Hermoni K, Khanin M, Danilenko M. (2015) The anti-cancer effects of carotenoids and other phytonutrients resides in their combined activity. , Arch. Biochem. Biophys 572, 28-35.
- 12.Shen K H, Hung J H, Chang C W. (2017) Solasodine inhibits invasion of human lung cancer cell through downregulation of miR-21 and MMPs expression. , Chemico-Biological Interactions 268, 129-135.
- 13.Bhaskar B, Ramesh K P. (2015) Genetically modified (GM) crop face an uncertain future in India: Bt Brinjal Appraisal – A perspective. , Annals of Plant Sciences 4, 960-975.
- 14.S N Chinedu, A C Olasumbo, O K Eboji, O C Emiloju, O K Arinola et al. (2011) Proximate and phytochemical analyses of Solanum aethiopicum L. and Solanum macrocarpon L. Fruits Res. , J. Chem. Sci 3, 63-71.
- 15.Nimenibo U, Omotayo R. (2019) Comparative proximate, mineral and vitamin composition of Solanum aethiopicum and Solanum melongenaNISEB. , Journal 3, 1-17.
- 16.T K Lim. (2013) Solanum aethiopicum Edible Medicinal and Non-medicinal Plants. , Dordrecht 56-78.
- 17.C N Ekweogu, V C Ude, Nwankpa P, Emmanuel O, E A Ugbogu. (2020) Ameliorative effect of aqueous leaf extract of Solanum aethiopicum on phenylhydrazine-induced anaemia and toxicity in rats Toxicol Res. 36, 227-238.
- 18.Afshari F, Serah H, Hashemi Z S. (2016) The cytotoxic effects of eggplant peel extract on human gastric adenocarcinoma cells and normal cells. , Mod Med Lab J 1, 42-48.
- 19.Tajik N, Tajik M, Mack I. (2017) The potential effects of chlorogenic acid, the main phenolic components in coffee, on health: a comprehensive review of the literature. , Eur. J. Nutr 56, 2215-2244.
- 20.Fraikue F B. (2016) Unveiling the potential utility of eggplant: a review. Conference Proceedings of INCEDI 883-895.
- 21.Plazas M, Lopez-Gresa M P, Vilanova S. (2013) Diversity and relationships in key traits 38 for functional and apparent quality in a collection of eggplant: fruit phenolics content, antioxidant activity, polyphenol oxidase activity, and browning. , J Agr Food Chem 61, 8871-8879.
- 22.Ahmed F A, Mubassara S, Sultana T. (2016) Phytoconstituents, bioactivity and antioxidant potential of some commercial brinjal (Solanum melongena L.) cultivars of Bangladesh. , Jahangirnagar University Journal of Biological Sciences 5, 41-50.
- 23.A M Salamatullah, M A Ahmed, M S Alkaltham, Hayat K, N S Aloumi et al. (2021) . Effect of Air-Frying on the Bioactive Properties of Eggplant (Solanum melongena L.). Processes 9, 435.
- 24.Im K, Lee J Y, Byeon H. (2016) In Vitro antioxidative and anti-inflammatory activities of the ethanol extract of eggplant (Solanum melongena) stalks in macrophage RAW 264.7 cells. Food Agr. , Immunol 27, 758-771.
- 25.Naem M Y, Ozgen S. (2020) Nutritional Content and Health Benefits of. , Eggplant.Turkish Journal of Agriculture - Food Science and Technology 7, 31-36.
- 26.Meyer R S, Bamshad M, Fuller D Q. (2014) Comparing medicinal uses of eggplant and related Solanaceae in China, India, and the Philippines suggests the independent development of uses, cultural diffusion, and recent species substitutions. , Economic Botany 68, 137-152.
- 27.Ghosh D, Konishi T. (2007) Anthocyanins and anthocyanin-rich extracts: role in diabetes and eye function. , Asia Pac J Clin Nutr 16, 200-208.
- 28.National Research Council. (2011) Committee for the Update of the Guide for the Care and Use of Laboratory Animals (2011): Guide for the Care and Use of Laboratory Animals. 8th edition. Washington (DC): National Academies Press (US);.
- 29.Friedewald W T, Levy R I, Fredrickson D S. (1972) Estimation of the concentration of low- density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. , Clin Chem 18, 499-502.
Cited by (4)
This article has been cited by 4 scholarly works according to:
Citing Articles:
BIO Web of Conferences (2024) OpenAlex
BIO Web of Conferences (2024) Crossref
